 This type of fire extinguisher has the added advantage of also cooling the fuel. Once this part of the fire triangle has been removed the fire is extinguished. They work by displacing the air (oxygen) from the area surrounding the fire. Carbon dioxide is non-polar as its dipoles cancel, and this contrasts with the polar molecule sulfur dioxide (SO 2).Ĭarbon dioxide fire extinguishers contain highly pressurized carbon dioxide which is a non-flammable inert gas, which acts as a smothering material. This is why it has a very low melting point 217 K (-56☌) at 5.2 atmospheres. Solid carbon dioxide has weak forces (van der Waals) between molecules which holds them together. It is an unusual solid as it sublimes (turning from solid to gas without going through the liquid state). 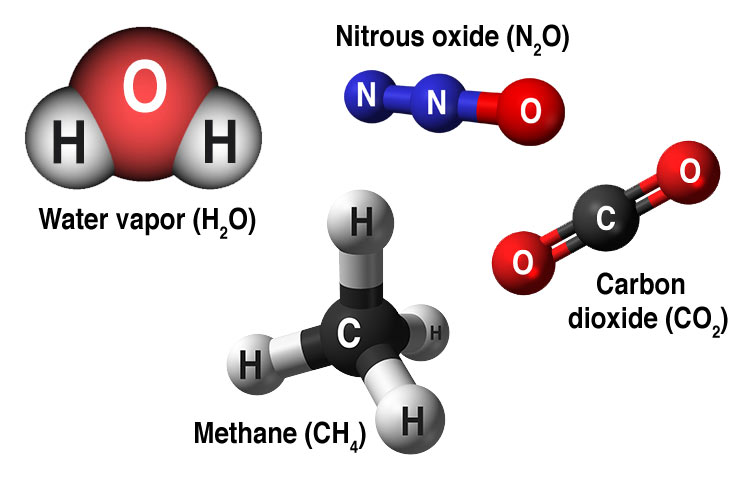
Solid carbon dioxide is known by chemists as cardice and by everyone else as dry ice. This gas was 'discovered' by Scottish scientist (physicist and chemist) Joseph Black (right). Carbon dioxide is a linear molecule with a bond angle of 180°. At room temperature carbon dioxide is a colourless gas which has a slightly sweet smell. Carbon dioxide has the formula CO 2 and at the centre of this linear molecule is a carbon atom joined by two pairs of double-bonds to the oxygen atoms, i.e O=C=O. 
NOTE: The GWP values were changed in 2007. The values in the Intergovernmental Panel on Climate Change (IPCC) Fourth Assessment Report ( AR4) in 2007 where refined from the IPCC Second Assessment Report ( SAR) values used previously and still in much of the literature.Carbon Dioxide - Molecule of the Month - May 2012 - HTML-only versionĬarbon Dioxide The gas we exhale that's both a Greenhouse gasĬarbon dioxide is a simple covalent molecule that most people have heard about, as it is often in the news due to its role in global warming. Therefore, releasing ONE KILOGRAM of sulphur hexafluoride is about equivalent to driving 5 cars for a year! (2) Greenhouse Gas The table shows that a release on 1 kg of this gas is equivalent to 22,800 kg or 22.8 tonnes of CO 2. The column on the right shows how much that chemical would warm the earth over a 100-year period as compared to carbon dioxide.įor example, sulphur hexafluoride is used to fill tennis balls. (1)Ĭlick here to download an expanded PDF table: GHG Lifetimes and GWPs (144 kB) How to read this table The following table shows the 100-year global warming potential for greenhouse gases reported by the United Nations Framework Convention on Climate Change (UNFCCC). Many of these compounds also have a depleting effect on ozone in the upper atmosphere. There are a wide variety of uses for SF 6, HFCs, and PFCs but they have been most commonly used as refrigerants and for fire suppression. These are sulphur hexafluoride (SF 6), hydrofluorocarbons (HFCs), and perfluorocarbons (PFCs). There are other greenhouse gases which have far greater global warming potential (GWP) but are much less prevalent. 
Water vapour is not considered to be a cause of man-made global warming because it does not persist in the atmosphere for more than a few days. It’s 20-year and 100-year GWP are basically the same. Nitrous oxide persists in the atmosphere for more than a century. Releasing 1 kg of N 2O into the atmosphere is about equivalent to releasing about 298 kg of CO2. The 100-year GWP is used to derive CO 2e. Methane’s 100-year GWP is about 28x CO2 – but it only persists in the atmosphere for a little more than a decade. Releasing 1 kg of CH4 into the atmosphere is about equivalent to releasing 84 kg of CO 2. All this time, it will be contributing to trapping heat and warming the atmosphere. 1 x – carbon dioxide (CO 2) NOTE: Any carbon dioxide added to the atmosphere will hang around for a long time: between 300 to 1,000 years.The three main greenhouse gases (along with water vapour) and their 20-year global warming potential (GWP) compared to carbon dioxide are: ( 1) 
Charts and tables in this Emissions section of our website convert all greenhouse gas (GHG) emissions into CO 2 equivalents so they can be compared.Įach greenhouse gas (GHG) has a different global warming potential (GWP) and persists for a different length of time in the atmosphere. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
